Main navigation
Procedure for Unclogging the LSR Sample Injection Port(or when instrument won’t take data)
Note: you’ll need to bring one extra test tube in order to perform some of the following procedures
PDF version of this procedure
- If you are the first person to use the instrument after power up and you see no evidence of signal (can’t see any dots on a dot plot), then do the following –
a. Place the DiVa software in Acquisition mode by clicking on the Acquire button (so the instrument will show data on the dot plots and histograms)
b. Place a tube with ~1 ml of DI H2O on the LSR at the sample injection port (SIP)
c. Push the Prime button
d. Watch the tube of DI H2O (you should see a rapid sequence of bubbles emitted from the end of the SIP, if you don’t see bubbles you’ll need to perform the procedure at section 2j)
e. Push the Run button
f. Observe the forward scatter (FSC-A) vs side scatter (SSC-A) dot plot while in Acquisition mode
g. If no dots show up in the FSC-A vs SSC-A plot during or after the Prime sequence, then the instrument CPU needs to be rebooted (the instrument CPU and PC are not communicating correctly)
h. Push the Standby button
i. Quit the DiVa software
j. Double click on the TeraTerm icon on the desktop
k. Once TeraTerm has started (black window shows up), hit the Enter key (an arrow icon will show up that looks like this “->”)
l. Type reboot and hit the Enter key
m. Once the reboot process starts, you’ll see scrolling lines of commands in the TeraTerm window (the reboot process may take a few minutes to start)
n. Wait for the reboot process to halt at the following line - “Cytometer IP Address: 192.168.1.1” (it may take several minutes to get to this point)
o. Restart the DiVa software (double click on the DiVa icon and login)
p. Wait for DiVa to “connect” (this can take several minutes)
q. Try running samples again
r. If you still cannot acquire data, proceed to the next steps
- If you have been running samples, but the instrument will no longer acquire data, then the sample injection port (SIP) is most likely plugged with sticky or aggregated cells.
a. Place a tube with ~1 ml of DI H2O on the instrument at the sample injection port (SIP)
b. Push the Prime button
c. Watch the tube of DI H2O (you should see a rapid sequence of bubbles emitted from the end of the SIP)
d. Push the Run button
e. Repeat steps b, c, and d, one more time
f. If you saw bubbles, then try running samples again, if you saw no bubbles, skip to section 2j
g. If samples still run slow or not at all, then put ~1 ml of Contrad in a test tube and run it on High for 5 minutes (the Contrad squeeze bottle is located on the wooden shelf near the hallway entrance door), repeat steps b, c, and d to clear debris loosened by the Contrad
h. Place a tube of ~2 ml of DI H2O on the instrument and run for 1 minute (to clear the Contrad from the system)
i. Try running samples again
j. If you saw no bubbles when running the Prime cycle, then the SIP is plugged solid
k. Place a tube with ~1 ml of DI H2O on the instrument and push the Standby button
l. Locate the tube of wire styluses in the bottom drawer below the LSR optical filter drawers

m. Remove one stylus

n. Remove the tube of DI H2O (leave silver SIP arm to one side, see photo below)
o. Remove the outer SIP tubing (see photos) by unscrewing the gray knurled round nut that is tapered on its lower end (located at the top of the silver outer SIP tube, not the large 1” in diameter black knurled round nut at the very top of the SIP) and sliding the outer tubing down and off the inner tubing. When removing the outer SIP tubing, prevent the BAL seal from coming loose.

p. Using the H2O squeeze bottle, put the tip of the squeeze bottle against the bottom end of the outer SIP tubing that was removed and squeeze several milliliters of H2O through the tubing, catching the water in the plastic tub provided at the LSR. This will clean out sticky cells that may have formed a clump in the outer SIP tubing.
q. Insert the wire stylus into the lower end of the inner SIP tubing (this is difficult to do and may take several minutes to successfully perform because the hole in the end of the SIP is extremely small). The cartoon below is drawn to scale.
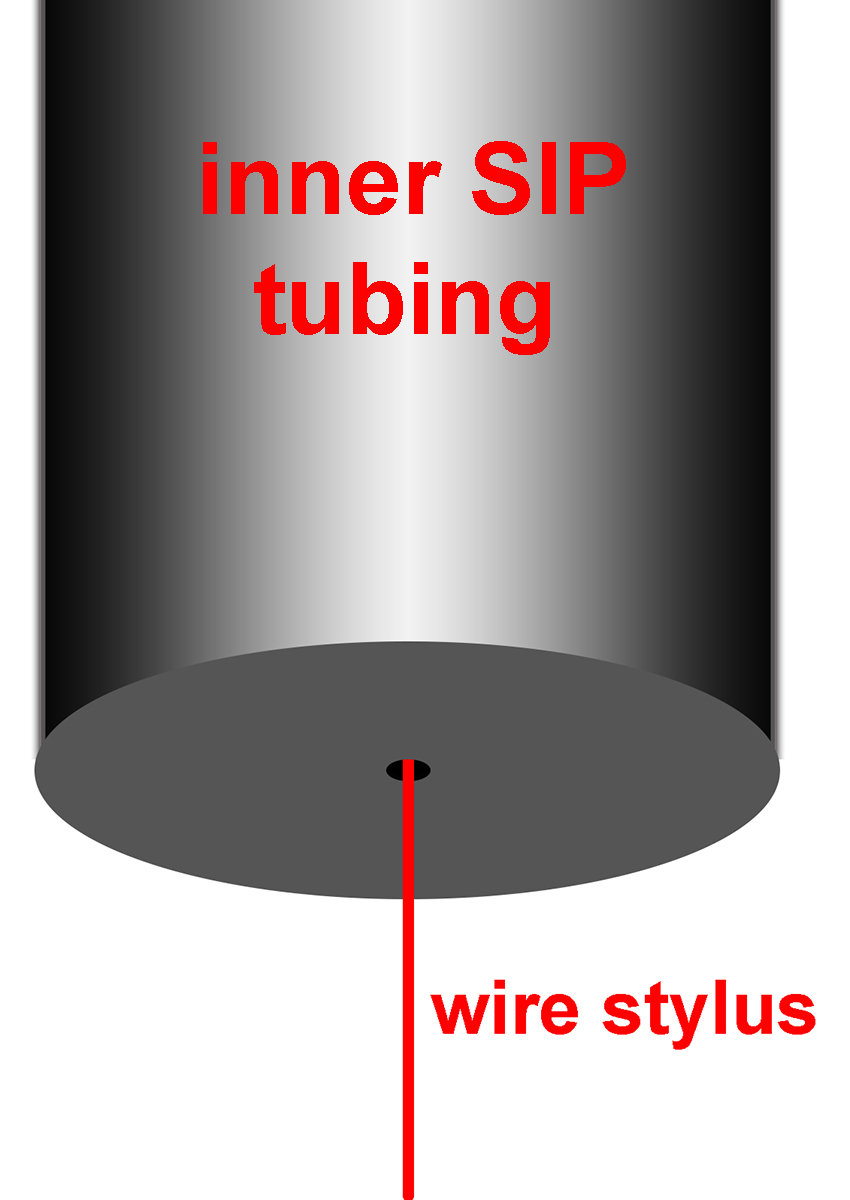
r. “Floss” the SIP tubing with the stylus by cycling it up and down in the SIP tubing (leave 1” of wire hanging out of the bottom of the SIP) ~5-10 cycles)
s. Remove the stylus
t. Place instrument in Run mode (push the Run button)
u. If the clog has been removed, the SIP should now drip
v. If it doesn’t drip, repeat the “flossing” steps q, r, and s
w. Once the SIP drips, replace the outer SIP tubing making sure to slide it up until it stops before tightening the knurled nut until it is snug (don’t over tighten the knurled nut))
x. Place ~1 ml of Contrad in a test tube and run it on High for 5 minutes
y. Place a tube of ~2 ml of DI H2O on the instrument and run for 1 minute
z. Try running samples again
If it is still not possible to acquire events, contact one of the Flow Cytometry staff.